Answers: 3
Other questions on the subject: English



Do you know the correct answer?
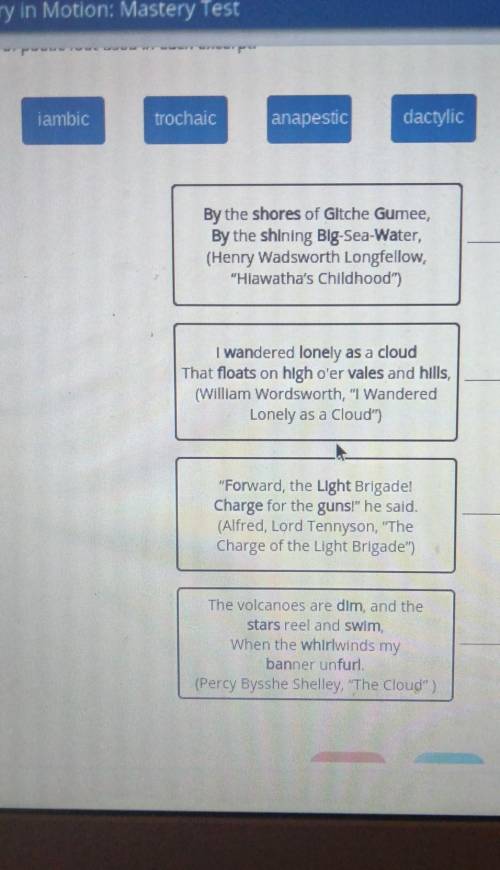
Identify the type of poetic foot used in each excerpt
...
...
Questions in other subjects:



Mathematics, 09.10.2021 02:40


Mathematics, 09.10.2021 02:40

History, 09.10.2021 02:40