Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:10, bettybales1986
What effect does nuclear radiation have on atoms?
Answers: 1


Chemistry, 22.06.2019 05:50, mayamabjishovrvq9
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2

Chemistry, 22.06.2019 15:30, dylannhandy
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2
Do you know the correct answer?
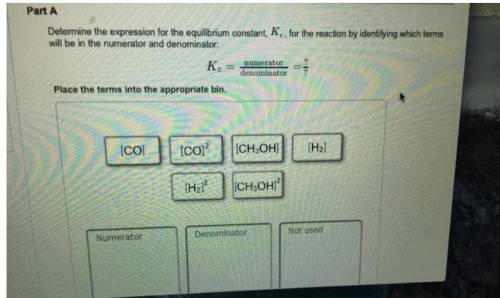
Determine the expression for the equilibrium constant, KcKcK_c, for the reaction by identifying whic...
Questions in other subjects:

Physics, 06.04.2020 23:35


Social Studies, 06.04.2020 23:35


Mathematics, 06.04.2020 23:35