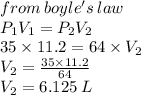Chemistry, 06.05.2021 21:00, angel213326
A gas occupies 11.2 L at a pressure of 35.0 mm . What is the volume of the gas if it is Increased to a pressure of 64.0 mm Hg?
A) 6.13 L
B) 110.2 L
C) 200.0 K
D) 20.5 L

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, jeffcarpenter
Write a net ionic equation to show that hydrofluoric acid, behaves as an acid in water.
Answers: 1

Chemistry, 22.06.2019 02:30, sotoamerica0814
98 ! and brainliest plz ! the below reaction can be categorized as more than one type of reaction. which reactions are these, and what are the types of reactions?
Answers: 1
Do you know the correct answer?
A gas occupies 11.2 L at a pressure of 35.0 mm . What is the volume of the gas if it is Increased to...
Questions in other subjects:

Physics, 30.06.2019 16:50



Business, 30.06.2019 16:50

Mathematics, 30.06.2019 16:50

History, 30.06.2019 16:50