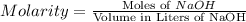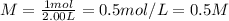Chemistry, 29.01.2020 02:55, 19thomasar
Achemist uses 0.25 l of 2.00 m h2so4 to completely neutralize a 2.00 l of solution of naoh. the balanced chemical equation of the reaction is given below. 2naoh + h2so4 na2so4 + 2h2o what is the concentration of naoh that is used?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1


Chemistry, 22.06.2019 22:30, StupidFatChipmunk
What must be in balance for temperatures to remain constant?
Answers: 1
Do you know the correct answer?
Achemist uses 0.25 l of 2.00 m h2so4 to completely neutralize a 2.00 l of solution of naoh. the bala...
Questions in other subjects:


English, 29.07.2019 12:10


Mathematics, 29.07.2019 12:10

Social Studies, 29.07.2019 12:10

History, 29.07.2019 12:10

English, 29.07.2019 12:10

Computers and Technology, 29.07.2019 12:10

Arts, 29.07.2019 12:10

Computers and Technology, 29.07.2019 12:10


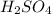 in 0.25 L of 2.00 M solution:
in 0.25 L of 2.00 M solution: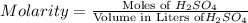
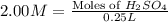
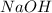 in 2.00 L of an unknown Molarity :
in 2.00 L of an unknown Molarity :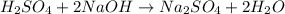
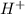 ions.
ions.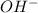 ions.
ions.