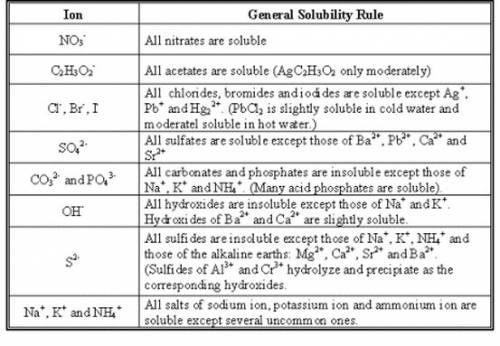
There are solubility rules for inorganic compounds in water as shown in the picture attached. For organic compounds, we use the concept of polarity.
1. NaCl: Since NaCl is a chloride and Na is not included in the list of insoluble, therefore, NaCl is highly soluble.
2. CH₃Cl: This compound is organic. Since the electonegativity between C and Cl is great, it tends to be polar. Because water is also polar, this is highly soluble.
3. CH₃OH: This is an alcohol. Alcohols with short chains of hydrocarbon are highly soluble in water because of hydrogen bonding (-OH).
4. KOH: From the table, KOH is highly soluble. In fact, this is a strong solid which readily dissociates in water.
5. C₆H₆ is the benzene ring. This is a nonpolar molecule, so you would expect this to be insoluble with water.
6. C₆H₁₄ is hexane which is a long chain of hydrocarbons. Because there is no point of hydrogen bonding, this is nonpolar, and therefore, insoluble in water.
7. KNO₃ is highly soluble because all nitrates are soluble.
8. CCl₄ is an organic compound. Individually, C-Cl bonds are polar. But because there are 4 of them, they cancel out. As a result, there is a balance of partial charges which makes it nonpolar. Thus, this is insoluble in water.
9. NaSO₄: from the table, this is highly soluble.
10. CH₃CH₂OH: Although it is composed of the nonpolar hydrocarbon chain, the OH group is much stronger such that it is highly soluble in water.
11. CH₂Cl₂: This is polar because there is an imbalance due to the C-H and C-Cl bonds. Thus, it is highly soluble in water.
12. CH₃COOH is a carboxylic acid which is even more soluble than alcohols. Thus, this is highly soluble in water.