
Chemistry, 19.01.2021 05:00, conniehodson
6.
Tristan completed a lab that combusted 5.0 grams of sodium
Write a complete balanced equation for the combustion of sodium.
a
b. What is the maximum amount of product that Tristan can obtain in this lab?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, pinkycupcakes3oxbqhx
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 16:30, ddmoorehouseov75lc
Correct relationship between molecular formula and empirical formula
Answers: 1

Chemistry, 22.06.2019 19:30, xxaurorabluexx
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2

Chemistry, 23.06.2019 00:30, tateandvioletAHS14AY
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
Do you know the correct answer?
6.
Tristan completed a lab that combusted 5.0 grams of sodium
Write a complete balanced equat...
Write a complete balanced equat...
Questions in other subjects:





Physics, 18.10.2019 10:10

Mathematics, 18.10.2019 10:10

Mathematics, 18.10.2019 10:10

History, 18.10.2019 10:10

Mathematics, 18.10.2019 10:10

Mathematics, 18.10.2019 10:10


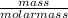
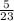 = 0.217moles
= 0.217moles 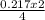 = 0.11mole of Na₂O
= 0.11mole of Na₂O 



