Calculate ΔH° = for the chemical reaction
Cl2(g) + F2(g) --->2ClF(g)
Bond Energies...

Chemistry, 17.12.2020 18:00, NycJahlilThegoat
Calculate ΔH° = for the chemical reaction
Cl2(g) + F2(g) --->2ClF(g)
Bond Energies, kJ mol-1
F---F 159
Cl--Cl 243
Cl--F 255

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, mpchop
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3


Chemistry, 22.06.2019 14:30, Cartucho1978
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1

Chemistry, 22.06.2019 19:00, andrecoral105
A4.86 g piece of metal was placed in a graduated cylinder containing 15.5 ml of water. the water level rose to 17.3 ml. what is the density of the metal. i need the steps of how to solve it to so i can use a formula to work out other problems.
Answers: 1
Do you know the correct answer?
Questions in other subjects:


History, 19.10.2019 16:10


Mathematics, 19.10.2019 16:10

Mathematics, 19.10.2019 16:10

Biology, 19.10.2019 16:10

History, 19.10.2019 16:10

Chemistry, 19.10.2019 16:10

English, 19.10.2019 16:10


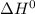 for the reaction
for the reaction 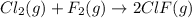 is -108kJ
is -108kJ![\Delta H=[n\times B.E_{reactants}]-[n\times B.E{products}]](/tpl/images/0995/4412/589d1.png)
![\Delta H=[1\times B.E_{Cl_2}+1\times B.E_{F_2}]-[2\times B.E_{ClF}]](/tpl/images/0995/4412/c74ae.png)
![\Delta H=[(1\times 243)+(1\times 159)]-[2\times 255]](/tpl/images/0995/4412/fbd29.png)





