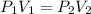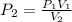Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3


Chemistry, 23.06.2019 01:00, MrTeriffic
Imagine if during the cathode ray experiment, the size of the particles of the ray was the same as the size of the atom forming the cathode. which other model or scientific observation would have also been supported? 1. this would support dalton's postulates that proposed the atoms are indivisible because no small particles are involved. 2. this would support bohr's prediction about electrons moving in orbits having specific energy. 3. this would support bohr's prediction about electrons being randomly scattered around the nucleus in the atom. 4. this would support dalton's postulates that proposed that atoms combine in fixed whole number ratios to form compounds.
Answers: 1
Do you know the correct answer?
If 489 mL of carbon dioxide has a pressure of 2.75 atm. If the gas is compressed to 254 mL, what is...
Questions in other subjects:

Mathematics, 04.12.2020 19:40


English, 04.12.2020 19:40

English, 04.12.2020 19:40

Mathematics, 04.12.2020 19:40

English, 04.12.2020 19:40



Mathematics, 04.12.2020 19:40

Biology, 04.12.2020 19:40