

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 14:30, malenacastillo4887
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3
Do you know the correct answer?
Calculate the molality of 25.0 grams of potassium bromide dissolved in 900 mL of pure water. (densit...
Questions in other subjects:






Mathematics, 16.11.2019 15:31




Biology, 16.11.2019 15:31


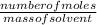
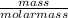
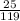 = 0.21mole
= 0.21mole 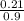 = 0.23mol/kg
= 0.23mol/kg



