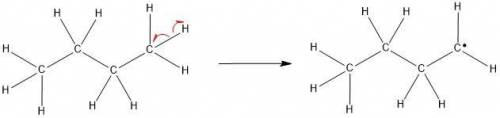
Chemistry, 20.07.2020 01:01, cheesecake1919
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG (Liquefied Petroleum Gas) cylinders (a common source of cooking gas). It has two arrangements of carbon atoms: a straight chain and a branched chain. Using this information, draw the structure of the tertiary butyl radical that will form upon removal of a hydrogen atom.
Requried:
Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. Include all free radicals by right-clicking on an atom on the canvas and then using the Atom properties to select the monovalent radical.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:00, boxergirl2062
Scientific evidence tells us that the cause of earths four season is the tilt of earth as it revolves around the sun. the student is instructed to illustrate this information in a science notebook. how will the student illiterate winter in the northern hemisphere?
Answers: 3


Chemistry, 22.06.2019 12:00, KKHeffner02
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1

Chemistry, 22.06.2019 14:00, asanchez4292
What type of matter is made of only one kind of atom
Answers: 2
Do you know the correct answer?
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG...
Questions in other subjects:


Mathematics, 05.08.2020 06:01


English, 05.08.2020 06:01




Mathematics, 05.08.2020 06:01

Mathematics, 05.08.2020 06:01