
Chemistry, 22.05.2020 21:09, antboozeoszxej
Consider the decomposition of water, is energy released or absorbed in this reaction? Discuss bond energy in your answer.!
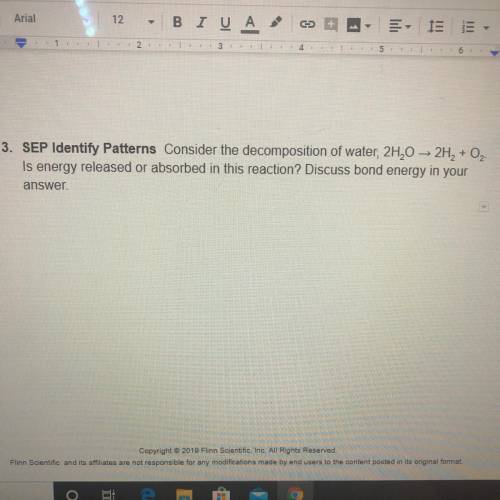

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:00, georgesarkes12
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 23.06.2019 12:30, marleacorey
All chemicals, even safe chemicals, such as sodium chloride( table salt), are toxic if exposure is high enough. (true or false)
Answers: 2

Chemistry, 23.06.2019 13:00, madelyngv97
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 23.06.2019 14:30, maelonramirez
William has eight more nickels than dimes in his pocket for a total of $2.50. which equation could be used to determine the number of x dimes in his pocket?
Answers: 1
Do you know the correct answer?
Consider the decomposition of water, is energy released or absorbed in this reaction? Discuss bond e...
Questions in other subjects:


Biology, 18.03.2022 17:10








Mathematics, 18.03.2022 17:20






