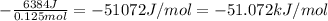Chemistry, 21.04.2020 22:31, cassidy100117
When 4.98 g of NaOH was dissolved in 52.79 g of water in a calorimeter at 23.7 oC, the temperature of the solution went up to 50.1 oC. What is the enthalpy change in kJ/mole of sodium hydroxide? Assume the specific heat of the mixture is the same as water.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:30, alaf05160
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks. energy was destroyed inside the blocks. energy was absorbed into the blocks from outside the system. energy was transferred from the warmer block to the cooler block.
Answers: 2

Do you know the correct answer?
When 4.98 g of NaOH was dissolved in 52.79 g of water in a calorimeter at 23.7 oC, the temperature o...
Questions in other subjects:











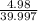 moles of NaOH = 0.125 moles of NaOH
moles of NaOH = 0.125 moles of NaOH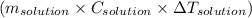 , where m is mass C is specific heat and
, where m is mass C is specific heat and  is change in temperature.
is change in temperature.![[57.77g\times 4.186\frac{J}{g.^{0}\textrm{C}}\times (50.1-23.7)^{0}\textrm{C}]](/tpl/images/0616/0525/cdabc.png)