Chemistry, 14.04.2020 21:08, Qwerty2771
A chemistry student weighs out 0.154 g of chloroacetic acid (HCH2CICO2) into a 250. mL volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.1400 M NaOH solution. Calculate the volume of NaOH solution the student will need to add to reach the equivalence point.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, tgraveslaylay2743
Bose-einstein condensation occurs at what temperature?
Answers: 2

Chemistry, 22.06.2019 22:30, darceline1574
Vi limitens. vastery test select the correct answer. which statement explains why large atoms are more reactive than small atoms? a. large atoms have valence electrons farther from the nucleus and lose them more readily. b. large atoms have greater ionization energy, which they can utilize during a reaction. c. large atoms have a greater number of electrons that they can lose during a reaction. d. large atoms have more energy levels, so they have more energy to pass on in a reaction. reset next
Answers: 3

Chemistry, 23.06.2019 01:10, dontcareanyonemo
Can someone check my work 98 5.05 acids and bases for this assignment you will be comparing acids and bases. the chart below will you organize the information needed: acids bases chemical properties (2) deodorant detergent vinger dish soap physical properties (2) orange juice toilet cleaner drain cleaner window cleaner ph level acid ph goes from 0-4 bases ph goes from 10-14 examples around you (2) vinger coffee lemon juice dark chocolate
Answers: 3

Chemistry, 23.06.2019 06:40, hackman1216
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants. thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction. ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch, are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
Do you know the correct answer?
A chemistry student weighs out 0.154 g of chloroacetic acid (HCH2CICO2) into a 250. mL volumetric fl...
Questions in other subjects:











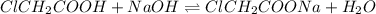
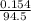 moles of chloroacetic acid
moles of chloroacetic acid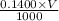 moles
moles