
Chemistry, 07.03.2020 01:18, kingbudd129
Calculate the pH of adding 20 mL of 1 M NaOH solution to 100 mL of a 1 M acetic acid (CH3COOH) solution and 880 mL of distilled water. Assume that the Ka of acetic acid is 1.8 * 10-5

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Do you know the correct answer?
Calculate the pH of adding 20 mL of 1 M NaOH solution to 100 mL of a 1 M acetic acid (CH3COOH) solut...
Questions in other subjects:

Mathematics, 25.11.2019 23:31



Biology, 25.11.2019 23:31

Biology, 25.11.2019 23:31

Engineering, 25.11.2019 23:31

Mathematics, 25.11.2019 23:31



Mathematics, 25.11.2019 23:31


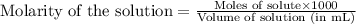
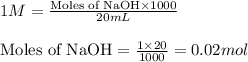
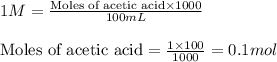
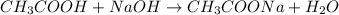
![pH=pK_a+\log(\frac{[salt]}{[acid]})](/tpl/images/0536/9084/e4eea.png)
![pH=pK_a+\log(\frac{[CH_3COONa]}{[CH_3COOH]})](/tpl/images/0536/9084/05ea7.png)
 = negative logarithm of acid dissociation constant of acetic acid = 4.74
= negative logarithm of acid dissociation constant of acetic acid = 4.74![[CH_3COONa]=\frac{0.020}{0.120}](/tpl/images/0536/9084/69ac9.png)
![[CH_3COOH]=\frac{0.080}{0.120}](/tpl/images/0536/9084/afc8b.png)






