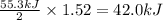A chemist measures the energy change during the following reaction: 1. This reaction is:. a. endothermic. b. exothermic. 2. Suppose 70.1 g of NO2 react. Will any heat be released or absorbed? A. Yes, absorbed. B. Yes, released. C. No. 3. If you said heat will be released or absorbed, calculate how much heat will be released or absorbed?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:00, kandi2565
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2

Chemistry, 22.06.2019 19:40, jholland03
What type of electromagnetic waves does the human eye see as the colors red blue or green a visible light waves b radio waves c infrared waves d microwaves
Answers: 1

Chemistry, 22.06.2019 23:10, carmenguabaoql9kv
Afusion reaction takes place between carbon and another element. neutrons are released, and a different element is formed. the different element is a) lighter than helium. b)heavier than helium. c)the same weight as helium. d)dependent on the element that reacted with carbon.
Answers: 3

Chemistry, 23.06.2019 01:00, stefaniethibodeaux
Substance 33°f 100°f peanut oil solid liquid margarine solid liquid chocolate chips solid liquid which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1
Do you know the correct answer?
A chemist measures the energy change during the following reaction: 1. This reaction is:. a. endothe...
Questions in other subjects:


Mathematics, 21.07.2021 18:00


Mathematics, 21.07.2021 18:00


Physics, 21.07.2021 18:00

English, 21.07.2021 18:00

Health, 21.07.2021 18:00



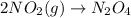
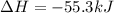 1. This reaction is:______. a. endothermic. b. exothermic.
1. This reaction is:______. a. endothermic. b. exothermic. 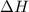 for Endothermic reaction is positive and
for Endothermic reaction is positive and 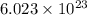 of particles.
of particles.
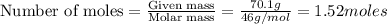
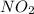 reacts, energy released = 55.3 kJ
reacts, energy released = 55.3 kJ