
Chemistry, 19.02.2020 05:41, levigabehart
A certain gas is present in a 15.0 LL cylinder at 2.0 atmatm pressure. If the pressure is increased to 4.0 atmatm the volume of the gas decreases to 7.5 LL . Find the two constants kiki, the initial value of kk, and kfkf, the final value of kk, to verify whether the gas obeys Boyle’s law by entering the numerical values for kiki and kfkf in the space provided.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:30, xxaurorabluexx
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2



Chemistry, 22.06.2019 23:40, tilievaughn14
What energy conversion occurs when a sling shot is used to shoot a rock across the room? (2 points) question 2 options: 1) stored mechanical energy is converted to mechanical energy. 2) stored mechanical energy is converted to radiant energy. 3) gravitational energy is converted to radiant energy. 4) gravitational energy is converted to mechanical energy.
Answers: 1
Do you know the correct answer?
A certain gas is present in a 15.0 LL cylinder at 2.0 atmatm pressure. If the pressure is increased...
Questions in other subjects:

Health, 14.07.2019 10:50

Biology, 14.07.2019 10:50

Mathematics, 14.07.2019 10:50


Social Studies, 14.07.2019 10:50


Mathematics, 14.07.2019 10:50

Health, 14.07.2019 10:50



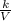
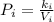
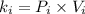
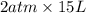
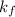 as follows.
as follows.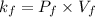
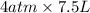
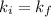 this means that it signifies that gas obeys boyle's law.
this means that it signifies that gas obeys boyle's law.



