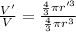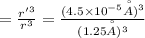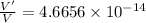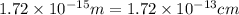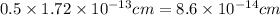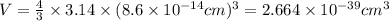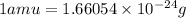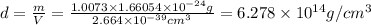An atom has a diameter of 2.50 Å and the nucleus of that atom has a diameter of 9.00×10−5 Å . Determine the fraction of the volume of the atom that is taken up by the nucleus. Assume the atom and the nucleus are a sphere.
fraction of atomic volume: ?
Calculate the density of a proton, given that the mass of a proton is 1.0073 amu and the diameter of a proton is 1.72×10−15 m.
density: ? g/cm^3

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, girly37
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3


Do you know the correct answer?
An atom has a diameter of 2.50 Å and the nucleus of that atom has a diameter of 9.00×10−5 Å . Determ...
Questions in other subjects:








English, 07.09.2020 03:01

Business, 07.09.2020 03:01


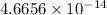 .
.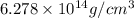 .
.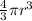
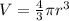 ..[1]
..[1]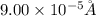

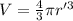 ..[2]
..[2]