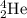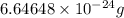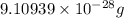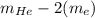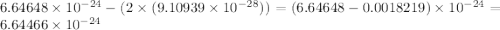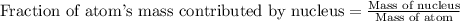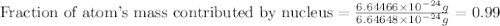Helium is the lightest noble gas and the second most abundant element (after hydrogen) in the universe. the mass of a helium−4 atom is 6.64648 × 10−24g, and each of its two electrons has a mass of 9.10939 × 10−28g. what fraction of this atom's mass is contributed by its nucleus?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, mimireds5419
1. combine iron and copper (ii) sulfate solution. (hint: iron will form the iron (iii) ion) fe + cuso4 → 2. combine lead (ii) nitrate and potassium iodide solutions. pb(no3)2+ kl → 3. combine magnesium metal and hydrochloric acid solution. mg + hcl → 4. electrolysis (splitting) of water. h2o → 5. burning magnesium. mg + o2 →
Answers: 3

Chemistry, 22.06.2019 08:00, danielhall
Identify a strong intermolecular force of attraction between an alcohol
Answers: 1

Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 23.06.2019 00:30, zaniathomasel
What is bromine+calcium iodide--> calcium bromide +iodine balanced
Answers: 1
Do you know the correct answer?
Helium is the lightest noble gas and the second most abundant element (after hydrogen) in the univer...
Questions in other subjects:

Mathematics, 07.12.2020 21:40

Computers and Technology, 07.12.2020 21:40

English, 07.12.2020 21:40

SAT, 07.12.2020 21:40