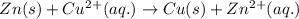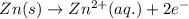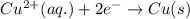Chemistry, 20.11.2019 17:31, fowers8376
Which species functions as the oxidizing agent in the following reduction-oxidation reaction:
zn(s) + cu^2+(aq) > cu(s) + zn^2+(aq)
a) zn^2+(aq)
b) zn(s)
c) cu^2+
d) cu(s)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:00, princessroyal
This graph gives information on changes in sea ice extent in the arctic ocean over a 30-year span. the overall trend shows in the ice extent. to address the trend, scientists need to ask themselves, one direct consequence of the trend is that
Answers: 1


Chemistry, 22.06.2019 15:30, dylannhandy
Using the first volume and temperature reading on the table as v1 and t1, solve for the unknown values in the table below. remember to use the rules of significant figures when entering your numeric response.
Answers: 2

Chemistry, 22.06.2019 16:00, ghadeeraljelawy
How does blood clotting prevent the entry of pathogens through cuts and wounds? answer asap,, this is due tomorrow. will mark as brainliest or whatever you call it : )
Answers: 2
Do you know the correct answer?
Which species functions as the oxidizing agent in the following reduction-oxidation reaction:
Questions in other subjects:











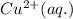 is the oxidizing agent for the given equation.
is the oxidizing agent for the given equation.