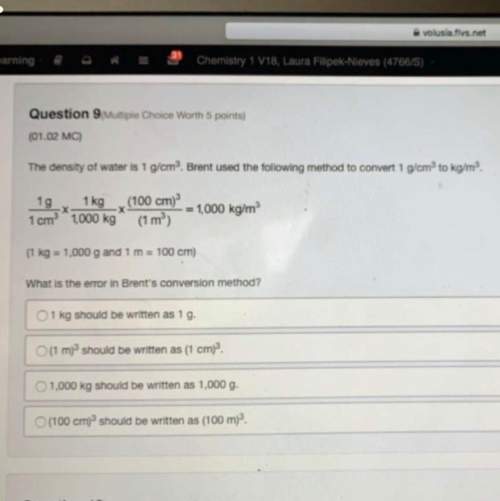
Phosphoric acid is a triprotic acid with the following pka values:
pka1=2.148, pka2=7.1...

Phosphoric acid is a triprotic acid with the following pka values:
pka1=2.148, pka2=7.198, pka3=12.375
you wish to prepare 1.000 l of a 0.0500 m phosphate buffer at ph 7.540. to do this, you choose to mix the two salt forms involved in the second ionization, nah2po4 and na2hpo4, in a 1.000 l volumetric flask and add water to the mark. what mass of each salt will you add to the mixture?
mass nah2po4
mass na2hpo4
what other combination of phosphoric acid and/or its salts could be mixed to prepare this buffer? (check all that apply).
h3po4 and nah2po4
h3po4 and na2hpo4
h3po4 and na3hpo4
nah2po4 and na3po4
na2hpo4 and na3po4

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:00, 767sebmont
Temperature and kinetic energy are proportional. a) adirectly b) directly c) indirectly
Answers: 2


Chemistry, 23.06.2019 11:50, michaelsoupios
How many moles of an ideal gas would occupy a 25.0 liter container when the temperature is 295 k and the pressure is 0.850 atm?
Answers: 2

Chemistry, 23.06.2019 14:00, Gaby702
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 6.00 mol fe and 8.45 mol nio(oh) react?
Answers: 1
Do you know the correct answer?
Questions in other subjects:


Biology, 08.01.2020 00:31



Computers and Technology, 08.01.2020 00:31