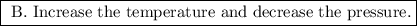** answer fast i will mark you brainliest and give 20 points * * this is a chemistry question so only answer if you are good at chemistry *
a certain reaction is endothermic in the forward direction and has a greater number of molecules on the product side. which set of conditions should be used to maximize the yield of the products?
a. decrease the temperature and decrease the pressure.
b. increase the temperature and decrease the pressure.
c. decrease the temperature and increase the pressure.
d. increase the temperature and increase the pressure.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3


Do you know the correct answer?
** answer fast i will mark you brainliest and give 20 points * * this is a chemistry question so on...
Questions in other subjects:



Mathematics, 31.03.2020 23:34

Mathematics, 31.03.2020 23:34

Mathematics, 31.03.2020 23:34

History, 31.03.2020 23:34

Chemistry, 31.03.2020 23:34



Chemistry, 31.03.2020 23:34