c) 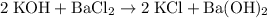
Explanation
Start by finding the formula for each of the four species.
The question is asking for a chemical equation. What that means that all species in this equation shall be neutral chemical compounds. Charges from ions shall cancel out within each species such that they end up appearing having no electrical charge.
Potassium hydroxide contains
Potassium ions
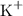
of charge +1, andHydroxide ions
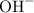
of charge -1.
Each potassium ion pairs with one hydroxide ion for their charge to cancel out. Potassium hydroxide shall therefore have a chemical formula of 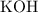 .
.
Barium chloride contains
Barium ions
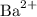
of charge +2, andChloride ions
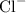
of charge -1.
It takes two chloride ions to cancel out the positive charge on a single barium ion. Barium chloride shall therefore have a chemical formula of 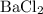 (note the subscript attached to chlorine.)
(note the subscript attached to chlorine.)
Similarly,
Potassium chloride would be written as
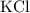
, andBarium hydroxide would be written as
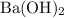
.
Hence the NOT YET BALANCED equation for this reaction:
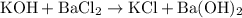 .
.
Atoms are conserved in chemical processes. The number of atoms of each element on the left hand side of the arrow shall thus equals to that on the right hand side.
Start by assigning a coefficient of one to one of the species. Then assign coefficients to others on both sides of the equation. It might be necessary to multiply all coefficients by a constant to eliminate any fractional coefficients.
Taking potassium hydroxide as an example:
Assuming that one formula unit of potassium hydroxide takes part in this reaction. There would be one potassium atom on the left hand side of the equation, which- by conservation- should also be present on the other side. One formula unit of potassium chloride should thus be found on the product side.
The chloride atom in potassium chloride attributes to 1/2 formula unit of barium chloride on the product side. There are thus 1/2 barium atoms on both sides of the equation. The chemical equation would therefore be:
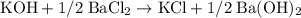
Multiply all coefficients by 2 to eliminate fractions. Hence the BALANCED chemical equation:
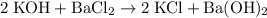 .
.
















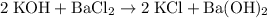
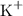 of charge +1, andHydroxide ions
of charge +1, andHydroxide ions 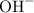 of charge -1.
of charge -1.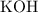 .
.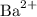 of charge +2, andChloride ions
of charge +2, andChloride ions 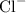 of charge -1.
of charge -1.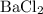 (note the subscript attached to chlorine.)
(note the subscript attached to chlorine.)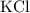 , andBarium hydroxide would be written as
, andBarium hydroxide would be written as 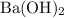 .
.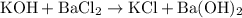 .
.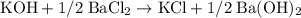
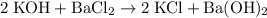 .
.



