
Chemistry, 19.09.2019 04:30, friendsalwaysbae
Homework 3 write the balanced equations. calculate how many grams of each reactant will be needed to obtain 100.0 grams of the insoluble product formed in the reaction. show complete solutions. 1. aluminum chloride + calcium hydroxide aluminum hydroxide + calcium chloride 2. mercury (ii) oxide mercury + oxygen 3. barium nitrate + copper (ii) sulfate barium sulfate+ copper (ii) nitrate 4. lead (ii) chloride + potassium iodide lead (ii) iodide + potassium chloride 5. sodium sulfide + copper (ii) chloride copper (ii) sulfide + sodium chloride

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, jusicca1109
100 points answer quick the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 07:30, SchoolFirst9811
The scheme below is from a series of reactions that are part of a synthesis of vitamin a. answer the following questions with reference to this scheme. (i) what is "reagent a"? (ii) draw a step-by-step mechanism which explains the formation of compound c from compound b (iii) which reagents would you use to form compound e from compounds c and d (reagents b and c)? for each reagent suggested above in (ii) explain the role of the reagent in the reaction to (iv) form compound e. you may wish to do this by drawing a mechanism. 1. addition of reagent a но reagent a 2. н, о" thо oh нон-с compound a. compound b. compound c .ch-оh 1. reagent b "сно 2. reagent c сh oh compound e. compound d.
Answers: 2

Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
Do you know the correct answer?
Homework 3 write the balanced equations. calculate how many grams of each reactant will be needed to...
Questions in other subjects:




Mathematics, 27.07.2019 06:50





English, 27.07.2019 06:50

History, 27.07.2019 06:50


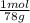 = 1,282 moles of Al(OH)₃.
= 1,282 moles of Al(OH)₃.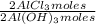 = 1,282 moles of AlCl₃ ×
= 1,282 moles of AlCl₃ × 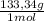 = 170,9 g of AlCl₃
= 170,9 g of AlCl₃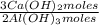 = 1,923 moles of Ca(OH)₂ ×
= 1,923 moles of Ca(OH)₂ × 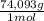 = 142,5 g of Ca(OH)₂
= 142,5 g of Ca(OH)₂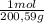 = 0,4985 moles of Hg.
= 0,4985 moles of Hg.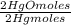 = 0,4985 moles of HgO ×
= 0,4985 moles of HgO × 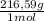 = 108,0 g of HgO
= 108,0 g of HgO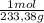 = 0,4285 moles of BaSO₄.
= 0,4285 moles of BaSO₄.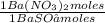 = 0,4285 moles of Ba(NO₃)₂ ×
= 0,4285 moles of Ba(NO₃)₂ × 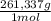 = 112,0 g of Ba(NO₃)₂
= 112,0 g of Ba(NO₃)₂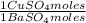 = 0,4285 moles of CuSO₄ ×
= 0,4285 moles of CuSO₄ × 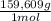 = 68,39 g of CuSO₄
= 68,39 g of CuSO₄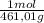 = 0,2169 moles of PbI₂.
= 0,2169 moles of PbI₂.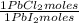 = 0,2169 moles of PbCl₂ ×
= 0,2169 moles of PbCl₂ × 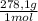 = 60,32 g of PbCl₂
= 60,32 g of PbCl₂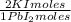 = 0,4338 moles of KI ×
= 0,4338 moles of KI × 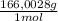 = 72,01 g of KI
= 72,01 g of KI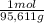 = 1,046 moles of CuS.
= 1,046 moles of CuS.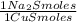 = 1,046 moles of Na₂S ×
= 1,046 moles of Na₂S × 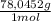 = 81,63 g of Na₂S
= 81,63 g of Na₂S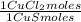 = 1,046 moles of CuCl₂ ×
= 1,046 moles of CuCl₂ × 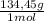 = 140,6 g of CuCl₂
= 140,6 g of CuCl₂



