
Chemistry, 09.07.2019 17:30, 1hannacarson
Consider the combustion of h2(g) 2h2(g)+o2(g)→2h2o(g). if hydrogen is burning at the rate of 0.49 mol/s, what is the rate of consumption of oxygen

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:10, vapelordcarl69
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 09:00, alydiale584
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1


Chemistry, 23.06.2019 03:00, amberskids2
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2
Do you know the correct answer?
Consider the combustion of h2(g) 2h2(g)+o2(g)→2h2o(g). if hydrogen is burning at the rate of 0.49 mo...
Questions in other subjects:











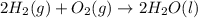
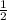 × 0.49 mol/s
× 0.49 mol/s



