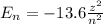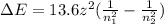How does quantum theory explain the emission spectra of atoms? a. electrons absorb the energy they need to jump to a higher energy level from any light source. b. a single, specific amount of energy is associated with each movement of an electron between energy levels. c. electrons gain and release energy in a slow, continuous fashion. d. the movement of an electron between energy levels occurs gradually as small amounts of energy are absorbed.

Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 05:10, adrianaglass12
A- a of 8.00 cm a of 34.0 nc. a , -, a of 25.0 nc is on of a of 15.0 cm .be toto aof ? a) b) c) j d) e)
Answers: 1

Physics, 22.06.2019 18:10, victoriakraus6599
A200-n force is applied to the foot-operated air pump. the return spring s exerts a 2.6-n·m moment on member oba for this position. determine the corresponding compression force c in the cylinder bd. if the diameter of the piston in the cylinder is 40 mm, estimate the air pressure generated for these conditions. state any assumptions. enter a positive number for the compression force c.
Answers: 2

Physics, 22.06.2019 21:10, tednequamoore4307
Aspecified volume of space contains an electric field for which the magnitude is given by e=e0cos(ωt). suppose that e0 = 20 v/m and ω = 1.0 × 107 s−1. part a what is the maximum displacement current through a 0.80 m2 cross-sectional area of this volume? express your answer with the appropriate units. idisp, max i d i s p , m a x = nothing nothing request answer part b how would this area have to be oriented relative to the electric field to get this maximum displacement current?
Answers: 2

Do you know the correct answer?
How does quantum theory explain the emission spectra of atoms? a. electrons absorb the energy they...
Questions in other subjects:

Geography, 22.03.2021 04:40

Mathematics, 22.03.2021 04:40





Mathematics, 22.03.2021 04:40


Mathematics, 22.03.2021 04:40