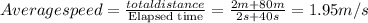1.the balanced chemical equation is as below
cu + 2 ag(no)₃ → cu(no₃)₂ +2 ag
2.the much silver produced is 50.9 g
step 1: find the moles of cu
moles = mass÷ molar mass
from periodic table the molar mass of cu = 63.5 g/mol
moles is therefore = 15.00 g÷ 63.5 g/mol = 0.236 moles
step 2: use the mole ratio to determine the moles of ag
from equation above cu: ag is 1: 2
therefore the moles of ag =0.236 moles x 2/1 =0.472 moles
step 3: find the mass of ag
mass = moles x molar mass
from periodic table the molar mass of ag =107.87 g/mol
mass = 0.472 moles x 107.87 g/mol = 50.9 g
3. the much silver collected worth is 1.797 oz
convert 50.9 g to oz
that is 1 gram = 0.0353 oz
50.9 grams =? oz
by cross multiplication
={(50.9 grams x 0.0353 oz) / 1 gram} =1.797 oz
















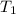 = 2 m
= 2 m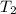 = 80 m m
= 80 m m