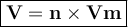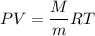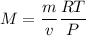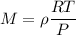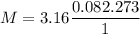The gas is Cl₂ - Chlorine gas
Further explanation
Stochiometry in Chemistry studies about chemical reactions, especially on quantitative, such as the calculation of volume, mass, amount, which is related to the number of ions, molecules, elements, etc.
In chemical calculations the reactants can be determined, the amount of substances that can be expressed in units of mass, volume, moles or determine a chemical formula for example the levels of substances or hydrate molecular formulas.
The volume per mole of a gas is called the molar volume
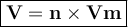
V = volume of gas, liters
n = mole
Vm = molar volume (liters / mol)
Molar volume depends on conditions:
1. Standard Conditions
Conditions at T 0 ° C and P 1 atm are stated by STP (Standard Temperature and Pressure).
PV = nRT
P = pressure, atm
V = volume of gas, liters
n = mole
R = constant = 0.082 L atm / mol K
T = 0 ° C = 273 ° K
So, at STP, Vm is 22.4 liters / mol.
2 Room Condition
Conditions at T 25 ° C and P 1 atm are stated by RTP (Room Temperature and Pressure). Vm in this condition = 24.4 liters / mol
From here we can input into the ideal gas equation to find an unknown value, molar mass (M) so that we know the gas
The density of a gas is 3.16 g / l at STP
for density of gas, The ideal gas equation becomes
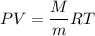
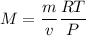
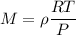
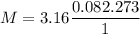
M = 70.74 g/mole
So the possible gas is Cl₂(chlorine) because the atomic mass of Cl is 35.5
Learn more
The mass of one mole of raindrops
link
moles of NaOH
link
moles of water you can produce
link
Keywords: gas, density, STP, molar mass