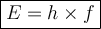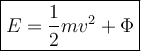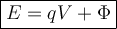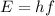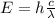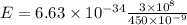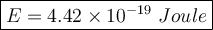The energy of individual photons of 450 nm light is 4.42 × 10⁻¹⁹ Joule
Further explanation
The term of package of electromagnetic wave radiation energy was first introduced by Max Planck. He termed it with photons with the magnitude is:
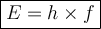
E = Energi of A Photon ( Joule )
h = Planck's Constant ( 6.63 × 10⁻³⁴ Js )
f = Frequency of Eletromagnetic Wave ( Hz )
The photoelectric effect is an effect in which electrons are released from the metal surface when illuminated by electromagnetic waves with large enough of radiation energy.
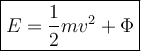
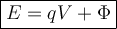
E = Energi of A Photon ( Joule )
m = Mass of an Electron ( kg )
v = Electron Release Speed ( m/s )
Ф = Work Function of Metal ( Joule )
q = Charge of an Electron ( Coulomb )
V = Stopping Potential ( Volt )
Let us now tackle the problem!
Given:
λ = 450 nm = 450 × 10⁻⁹ m
Unknown:
E = ?
Solution:
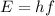
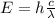
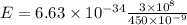
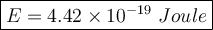
Learn morePhotoelectric Effect : linkStatements about the Photoelectric Effect : linkRutherford model and Photoelecric Effect : linkPhotoelectric Threshold Wavelength : linkAnswer details
Grade: High School
Subject: Physics
Chapter: Quantum Physics
Keywords: Quantum , Photoelectric , Effect , Threshold , Frequency , Electronvolt