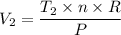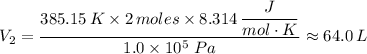Physics, 11.07.2021 18:00, puppylover72
When 2 moles of helium gas expand at a constant pressure p= 1.0×10^5 pascals, the temperature increase from 2 °c to 112 °c. If the initial volume of the gas was 45 liters. Cp= 20.8j/mol. K, Cv= 12.6j/mol. K. Determine i. The work done W by the gas as it exands

Answers: 3
Other questions on the subject: Physics

Physics, 21.06.2019 18:10, msmojangles
Amass has an acceleration of 10- 2t. what is its velocity and displacement if it started at x- 4m and the initial velocity was 8m/s? at what time does it have 0 velocity? graph all graphs.
Answers: 2


Physics, 22.06.2019 09:30, xxtonixwilsonxx
True or false graphs must include scales that increase by the same amount
Answers: 1

Do you know the correct answer?
When 2 moles of helium gas expand at a constant pressure p= 1.0×10^5 pascals, the temperature increa...
Questions in other subjects:




Mathematics, 07.12.2020 22:10


Social Studies, 07.12.2020 22:10

Mathematics, 07.12.2020 22:10


English, 07.12.2020 22:10

Mathematics, 07.12.2020 22:10