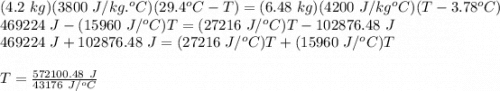An ice chest at a beach party contains 12 cans of soda at 3.78 °C. Each can of soda has a mass of 0.35 kg and a specific heat capacity of 3800 J/(kg C°). Someone adds a 6.48-kg watermelon at 29.4 °C to the chest. The specific heat capacity of watermelon is nearly the same as that of water. Ignore the specific heat capacity of the chest and determine the final temperature T of the soda and watermelon in degrees Celsius.

Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 11:00, bumpydumper
The dot diagram represents the motion of the motorcycle as it moves from left to right
Answers: 3

Physics, 22.06.2019 12:30, zayeboyd4971
Uppose we consider the system of the three capacitors as a single "equivalent" capacitor. given the charges of the three individual capacitors calculated in the previous part, find the total charge qtot for this equivalent capacitor. express your answer in terms of v and c.
Answers: 2

Physics, 22.06.2019 14:30, gabriellam20
A10nc charge sits at a point in space where the magnitude of the electric field is 1500 n/c. what will the magnitude of the field be if the 10 nc charge is replaced by a 20 nc charge? assume the system is big enough to consider the charges as small test charges.
Answers: 1

Physics, 22.06.2019 23:20, pr47723
Acharge q experiences no net force at a particular point in space. which of the following situations described below must always be true? -there are no other charges nearby. -if there are other charges nearby, they must all have the same sign as q. -if there are other charges nearby, they must all have the opposite sign of q. -if there are other charges nearby, the total positive charge must equal the total negative charge. -none of the above
Answers: 2
Do you know the correct answer?
An ice chest at a beach party contains 12 cans of soda at 3.78 °C. Each can of soda has a mass of 0....
Questions in other subjects:



Mathematics, 11.02.2021 23:50


Mathematics, 11.02.2021 23:50


Engineering, 11.02.2021 23:50


History, 11.02.2021 23:50

English, 11.02.2021 23:50


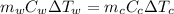
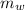 = mass of watermelon = 6.48 kg
= mass of watermelon = 6.48 kg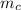 = mass of cans = (12)(0.35 kg) = 4.2 kg
= mass of cans = (12)(0.35 kg) = 4.2 kg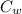 = specific heat capacity of watermelon = 3800 J/kg.°C
= specific heat capacity of watermelon = 3800 J/kg.°C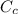 = specific heat capacity of cans = 4200 J/kg.°C
= specific heat capacity of cans = 4200 J/kg.°C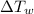 = Change in Temprature of watermelon = 29.4°C - T
= Change in Temprature of watermelon = 29.4°C - T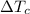 = Change in Temperature of cans = T - 3.78°C
= Change in Temperature of cans = T - 3.78°C