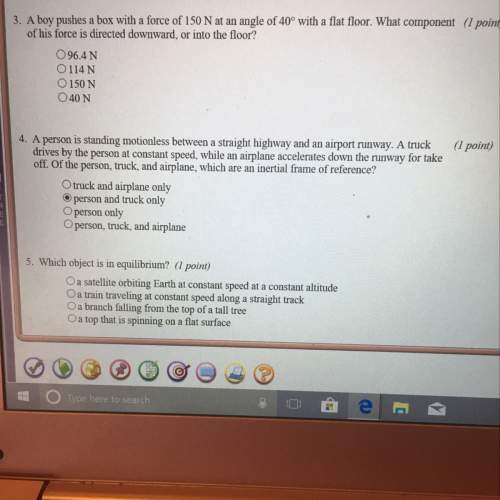
Physics, 13.05.2021 23:00, tiarafaimealelei
A 70 kg human body typically contains 140 g of potassium. Potassium has a chemical atomic mass of 39.1 u and has three naturally occurring isotopes. One of those isotopes, 40K (potassium), is radioactive with a half-life of 1.3 billion years and a natural abundance of 0.012 %. Each 40K (potassium) decay deposits, on average, 1.0 MeV of energy into the body. What yearly dose in Gy does the typical person receive from the decay of 40K (potassium) in the body? Express your answer using two significant figures.

Answers: 2
Other questions on the subject: Physics



Physics, 22.06.2019 14:30, gabriellam20
A10nc charge sits at a point in space where the magnitude of the electric field is 1500 n/c. what will the magnitude of the field be if the 10 nc charge is replaced by a 20 nc charge? assume the system is big enough to consider the charges as small test charges.
Answers: 1

Physics, 22.06.2019 15:00, banna01man
Greg drew a diagram to compare two of the fundamental forces. which labels belong in the areas marked x, y and z? gravitational force electromagnetic force x. attractive y repulsive z: infinite range x: infinite range y attractive z. repulsive x. always attractive y: infinite range z: attractive or repulsive x. attractive or repulsive y infinite range z: always attractive
Answers: 2
Do you know the correct answer?
A 70 kg human body typically contains 140 g of potassium. Potassium has a chemical atomic mass of 39...
Questions in other subjects:

Mathematics, 10.11.2019 11:31





Social Studies, 10.11.2019 11:31




Health, 10.11.2019 11:31