B


Answers: 2
Other questions on the subject: Physics

Physics, 22.06.2019 03:30, zahradawkins2007
As part of an industrial process, air as an ideal gas at 10 bar, 400k expands at steady state through a valve to a pressure of 4 bar. the mass flow rate of air is 0.5 kg/s. the air then passes through a heat exchanger where it is cooled to a temperature of 295k with negligible change in pressure. the valve can be modeled as a throttling process, and kinetic and potential energy effects can be neglected. (a) for a control volume enclosing the valve and heat exchanger and enough of the local surroundings that the heat transfer occurs at the ambient temperature of 295 k, determine the rate of entropy production, in kw/k. (b) if the expansion valve were replaced by an adiabatic turbine operating isentropically, what would be the entropy production? compare the results of parts (a) and (b) and discuss.
Answers: 3


Physics, 22.06.2019 23:00, shelbybibb99
Acommon technique in analysis of scientific data is normalization. the purpose of normalizing data is to eliminate irrelevant constants that can obscure the salient features of the data. the goal of this experiment is to test the hypothesis that the flux of light decreases as the square of the distance from the source. in this case, the absolute value of the voltage measured by the photometer is irrelevant; only the relative value conveys useful information. suppose that in part 2.2.2 of the experiment, students obtain a signal value of 162 mv at a distance of 4 cm and a value of 86 mv at a distance of 5.7 cm. normalize the students' data to the value obtained at 4 cm. (divide the signal value by 162.) then calculate the theoretically expected (normalized) value at 5.7 cm.
Answers: 2

Physics, 23.06.2019 00:30, struckedblazing
What is the speed vfinal of the electron when it is 10.0 cm from charge 1?
Answers: 1
Do you know the correct answer?
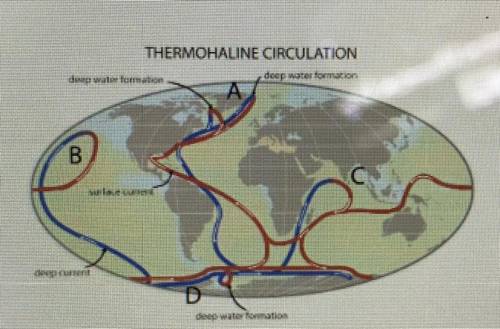
Which location on the map above is a source of North Atlantic deep water?
A
B
B
Questions in other subjects:


World Languages, 21.08.2019 05:40




History, 21.08.2019 05:40

Physics, 21.08.2019 05:40


Social Studies, 21.08.2019 05:40