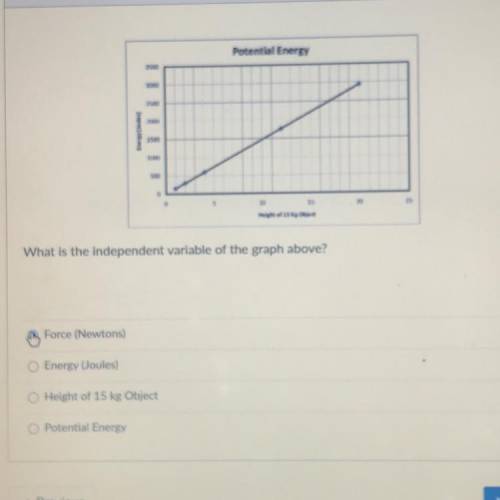
What is the independent variable of the graph above?
...

Answers: 2
Other questions on the subject: Physics

Physics, 21.06.2019 22:50, ijohnh14
If the temperature were raised very high, classically what would we expect the heat capacity per object to be for this one-dimensional system? give a numerical value. chigh t = __ j/k/object (one reason for the discrepancy is that the high-temperature limit assumes that the number of oscillators is large (n > > 1), which is not the case in this tiny system.)
Answers: 2


Physics, 22.06.2019 18:00, maren17
Air enters a gas turbine with two stages of compression and two stages of expansion at 100 kpa and 17°c. this system uses a regenerator as well as reheating and intercooling – the intercooler returns the air to the inlet temperature. the pressure ratio across each compressor is 4 ; 300 kj/kg of heat are added to the air in each combustion chamber; and the regenerator operates perfectly while increasing the temperature of the cold air by 20°c. determine the system’s thermal efficiency. assume isentropic operations for all compressor and the turbine stages and use constant specific heats at room temperature. (0.378)
Answers: 3

Physics, 22.06.2019 19:20, gokusupersaiyan12345
The dipole moment of the water molecule (h2o) is 6.17x10^-30 c. m. consider a water molecule located at the origin whose dipole moment p points in the +x-direction. a chlorine ion ( of charge-1.60x10^-19c , is located at x=3.00x10^-9m . assume that is much larger than the separation d between the charges in the dipole, so that the approximate expression for the electric field along the dipole axis can be used. a) find the magnitude of the electric force that the water molecule exerts on the chlorine ion. b) what is the direction of the electric force. -x-direction or +x-direction c) is this force attractive or repulsive?
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Biology, 21.07.2019 14:30


Social Studies, 21.07.2019 14:30

Social Studies, 21.07.2019 14:30

Biology, 21.07.2019 14:30


Mathematics, 21.07.2019 14:30

Mathematics, 21.07.2019 14:30