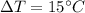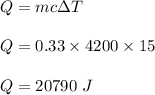Physics, 11.02.2021 14:00, valeriegarcia12
The can-chiller decreases the temperature of the liquid in the can by 15 °C.
The mass of liquid is 0.33 kg.
The specific heat capacity of the liquid is 4200 J / kg °C.
Calculate the energy transferred from the liquid as it cools.

Answers: 2
Other questions on the subject: Physics

Physics, 22.06.2019 00:40, camco301
Electroplating is a way to coat a complex metal object with a very thin (and hence inexpensive) layer of a precious metal, such as silver or gold. in essence the metal object is made the cathode of an electrolytic cell in which the precious metal cations are dissolved in aqueous solution. suppose a current of 480.ma is passed through an electroplating cell with an aqueous solution of agno3 in the cathode compartment for 46.0 seconds. calculate the mass of pure silver deposited on a metal object made into the cathode of the cell. round your answer to 3 significant digits. also, be sure your answer contains a unit symbol. ×10μ
Answers: 3

Physics, 22.06.2019 02:30, coltonstevens15
Which feature of a heating curve indicates a change ofn state
Answers: 1

Do you know the correct answer?
The can-chiller decreases the temperature of the liquid in the can by 15 °C.
The mass of liquid is...
Questions in other subjects:


English, 15.12.2020 18:10

Mathematics, 15.12.2020 18:10

Biology, 15.12.2020 18:10

Mathematics, 15.12.2020 18:10


English, 15.12.2020 18:10

English, 15.12.2020 18:10


History, 15.12.2020 18:10