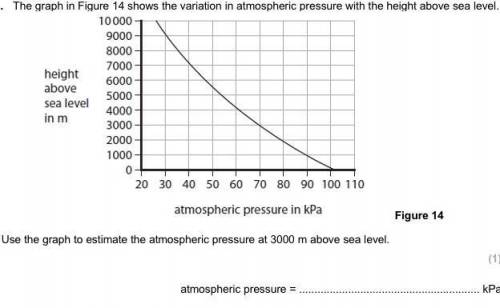
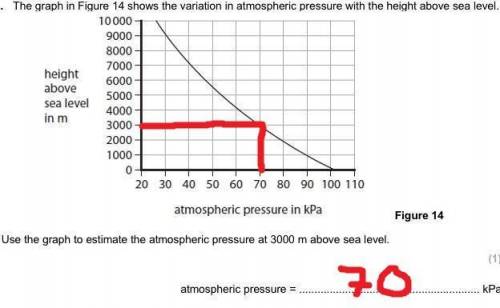
Yeah I need help because I don’t get this
...

Answers: 2
Other questions on the subject: Physics

Physics, 22.06.2019 05:50, kamrulh278
Acylinder with a movable piston contains 11.7 moles of a monatomic ideal gas at a pressure of 1.32×10^5 pa. the gas is initially at a temperature of 300 k. an electric heater adds 43200 j of energy into the gas while the piston moves in such a way that the pressure remains constant. cp=20.79 j k^−1 mol^−1 for a monatomic ideal gas, and that the number of gas molecules is equal to avogadro's number (6.022×10^23) times the number of moles of the gas. (a) what is the temperature of the gas after the energy is added? (b) what is the change in volume of the gas? (c) how much work is done by the gas during this process?
Answers: 3


Physics, 23.06.2019 02:30, pruittjr5161
Based on its type of chemical bond, which of the following has the highest boiling point? potassium chloride ethyl alcohol water ammonia
Answers: 1

Physics, 23.06.2019 11:40, edenlbarfield
As the ball rolls down the ramp, there are changes in kinetic and potential energy. how much kinetic energy does the ball have when it reaches point b? a) 0j b) 25j c) 50j d) 100
Answers: 3
Do you know the correct answer?
Questions in other subjects:

English, 30.03.2020 22:57


Mathematics, 30.03.2020 22:57





Health, 30.03.2020 22:57