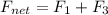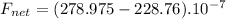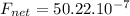Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 05:50, kamrulh278
Acylinder with a movable piston contains 11.7 moles of a monatomic ideal gas at a pressure of 1.32×10^5 pa. the gas is initially at a temperature of 300 k. an electric heater adds 43200 j of energy into the gas while the piston moves in such a way that the pressure remains constant. cp=20.79 j k^−1 mol^−1 for a monatomic ideal gas, and that the number of gas molecules is equal to avogadro's number (6.022×10^23) times the number of moles of the gas. (a) what is the temperature of the gas after the energy is added? (b) what is the change in volume of the gas? (c) how much work is done by the gas during this process?
Answers: 3

Physics, 22.06.2019 09:30, xxtonixwilsonxx
True or false graphs must include scales that increase by the same amount
Answers: 1

Physics, 22.06.2019 11:30, azduce
(2) (a) you have a simple circuit that consists of only a battery (δvbat=1.5v) and two resistors with resistance r1=10ω and r2=5ω, connected in series with each other. what is the current running through the battery? (b) you re-arrange your circuit so now r2 is attached in parallel to r1 rather than in series. what is the current running through the battery? (c) you add an additional resistor r3=7ω on the same branch as r2. what is the current running through the battery? (d) what is the power dissipated in r3?
Answers: 3
Do you know the correct answer?
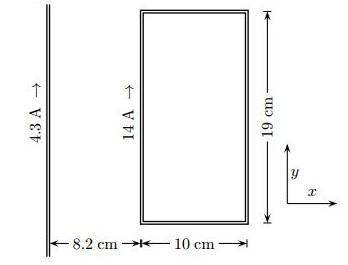
Find the magnitude of the net force exerted on the loop by the magnetic field created by the long wi...
Questions in other subjects:

Mathematics, 23.10.2021 14:00

History, 23.10.2021 14:00

History, 23.10.2021 14:00

Health, 23.10.2021 14:00

Social Studies, 23.10.2021 14:00

Chemistry, 23.10.2021 14:00

Mathematics, 23.10.2021 14:00


Mathematics, 23.10.2021 14:00

Mathematics, 23.10.2021 14:00


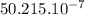 N
N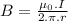
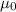 is permeability of free space and is
is permeability of free space and is 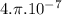 T.m/A
T.m/A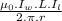
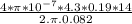
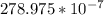 N
N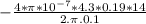
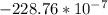 N
N