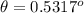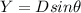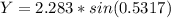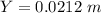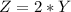Answers: 3
Other questions on the subject: Physics

Physics, 22.06.2019 05:50, kamrulh278
Acylinder with a movable piston contains 11.7 moles of a monatomic ideal gas at a pressure of 1.32×10^5 pa. the gas is initially at a temperature of 300 k. an electric heater adds 43200 j of energy into the gas while the piston moves in such a way that the pressure remains constant. cp=20.79 j k^−1 mol^−1 for a monatomic ideal gas, and that the number of gas molecules is equal to avogadro's number (6.022×10^23) times the number of moles of the gas. (a) what is the temperature of the gas after the energy is added? (b) what is the change in volume of the gas? (c) how much work is done by the gas during this process?
Answers: 3

Physics, 22.06.2019 12:10, aedelfrance9250
Aspring has a natural length of 8 m. if a 12-n force is required to keep it stretched to a length of 10 m, how much work w is required to stretch it from 8 m to 16 m? (round your answer to two decimal places.)
Answers: 1


Physics, 22.06.2019 13:40, mvongphakdy8746
Consider a double atwood machine constructed as follows: a mass 4m is suspended from a string that passes over a massless pulley on frictionless bearings. the other end of this string supports a second similar pulley, over which passes a second string supporting a mass of 3m at one end and m at the other. using two suitable generalized coordinates, set up the lagrangian and use the lagrange equations to find the acceleration of the mass 4m when the system is released. explain why the top pulley rotates even though it carries equal weights on each side.
Answers: 2
Do you know the correct answer?
You illuminate a slit with a width of 77.7 μm with a light of wavelength 721 nm and observe the resu...
Questions in other subjects:

Chemistry, 01.12.2020 14:00

History, 01.12.2020 14:00

Mathematics, 01.12.2020 14:00


Biology, 01.12.2020 14:00


English, 01.12.2020 14:00

Social Studies, 01.12.2020 14:00

Health, 01.12.2020 14:00

English, 01.12.2020 14:00


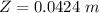
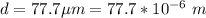
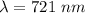
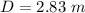
![\theta = sin ^{-1}[\frac{m \lambda}{d} ]](/tpl/images/0703/3846/42b61.png)
![\theta = sin ^{-1}[\frac{1 *721*10^{-9}}{ 77.7*10^{-6}} ]](/tpl/images/0703/3846/abce3.png)