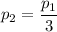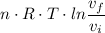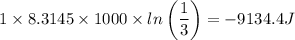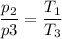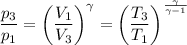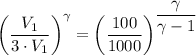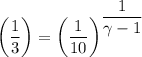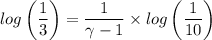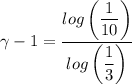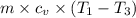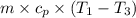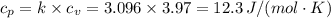Physics, 17.06.2020 05:57, tanyalynn12345
An ideal diatomic gas undergoes a cyclic process. In the first step, the gas undergoes an isothermal expansion from V1 to 3.00 V1. In the second step of the process the gas undergoes an isovolumetric decrease in pressure. In the third step the gas undergoes an adiabatic compression from 3.00 V1 back to V1 completing the cycle.
Required:
a. Sketch the cycle.
b. In terms of P., V. and T., determine P2, P3, T3.
c. In terms of P., V. To determine W, Q and ΔE int for each step. Take T, to be between 100K and 1000K

Answers: 3
Other questions on the subject: Physics

Physics, 22.06.2019 09:30, abdominguez7187
An electric clothes dryer has a resistance of 8 ohms. it draws 30 a of current. what is the voltage, in volts, of the wall outlet that it is plugged into?
Answers: 2

Physics, 22.06.2019 19:30, paigehixson342
Coal contains energy. a. light b. kinetic c. chemical d. mechanical
Answers: 1

Physics, 22.06.2019 21:30, carleygalloway103
Calculate the minimum energy required to remove one proton from the nucleus 126c. this is called the proton-removal energy. (hint: find the difference between the mass of a 126c nucleus and the mass of a proton plus the mass of the nucleus formed when a proton is removed from 126c.) express your answer with the appropriate units. emin e m i n = nothing nothing request answer part b how does the proton-removal energy for 126c compare to the binding energy per nucleon for 126c, calculated using eb=(zmh+nmn−azm)c2?
Answers: 1

Do you know the correct answer?
An ideal diatomic gas undergoes a cyclic process. In the first step, the gas undergoes an isothermal...
Questions in other subjects:


Mathematics, 06.03.2021 05:10

Mathematics, 06.03.2021 05:10



Mathematics, 06.03.2021 05:10


Mathematics, 06.03.2021 05:10