
The rate at which a metal alloy oxidizes in an oxygen-containing atmosphere is a typical example of the practical utility of the Arrhenius equation. For example, the rate of oxidation of a magnesium alloy is represented by a rate constant, k. The value of k at 300°C is 1.05 * 10-8kg/(m4 # s). At 400°C, the value of k rises to 2.95 * 10-4kg/(m4 # s). Calculate the activation energy, Q, for this oxidation process (in units of kJ/mol).

Answers: 3
Other questions on the subject: Physics

Physics, 22.06.2019 10:10, natalieagustinlop54
Henry, whose mass is 95 kg, stands on a bathroom scale in an elevator. the scale reads 830 n for the first 2.4 s after the elevator starts moving, then 930 n for the next 2.4 s. part a what is the elevator's speed 4.8 s after starting? express your answer with the appropriate units.
Answers: 2



Physics, 23.06.2019 01:30, ana2896
Hydroelectricity provides about 7 percent of the electricity generated in the united states. (a) explain how a hydroelectric power plant converts energy stored in water into electricity. (b) identify two factors that determine the amount of electricity that can be generated by an individual hydroelectricity power plant. (c) describe the two main types of land-based hydroelectric power plants. (d) describe two economic advantages and two environmental disadvantages of hydroelectricity.
Answers: 1
Do you know the correct answer?
The rate at which a metal alloy oxidizes in an oxygen-containing atmosphere is a typical example of...
Questions in other subjects:

Mathematics, 31.10.2019 11:31

Mathematics, 31.10.2019 11:31


Social Studies, 31.10.2019 11:31


Mathematics, 31.10.2019 11:31


Social Studies, 31.10.2019 11:31

Mathematics, 31.10.2019 11:31

English, 31.10.2019 11:31


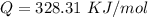
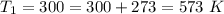
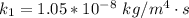
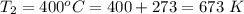
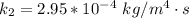
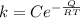
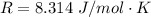
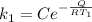
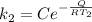
![\frac{k_1 }{k_2} = e^{(\frac{Q}{R} [\frac{1}{\frac{T_2 - 1}{T_1} } ] )}](/tpl/images/0668/9767/c32ab.png)
![ln [\frac{k_1}{k_2} ] = \frac{Q}{R} * [\frac{1}{\frac{T_2 -1}{T_1} } ]](/tpl/images/0668/9767/33b26.png)
![ln [\frac{1.05 *10^{-8}}{2.95 *10^{-4}} ] = \frac{Q}{8.314} * [\frac{1}{\frac{673 -1}{573} } ]](/tpl/images/0668/9767/45b57.png)




