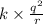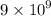Answers: 1
Other questions on the subject: Physics



Physics, 22.06.2019 10:50, milkshakegrande101
Asubject in a clinical research trial experiences a serious, unanticipated adverse drug experience. how should the investigator proceed, with respect to the irb, after the discovery of the adverse event occurrence? a. do not report the adverse drug experience to the irb since it is a common adverse experience. b. report the adverse drug experience to the irb only if there are several other occurrences. c. report the adverse drug experience as part of the continuing review report. d. report the adverse drug experience in a timely manner, in keeping with the irb's policies and procedures, using the forms or the mechanism provided by the irb.
Answers: 1

Physics, 22.06.2019 13:00, mandilynn22
Nacidified solution was electrolyzed using copper electrodes. a constant current of 1.18 a caused the anode to lose 0.584 g after 1.52 ✕ 103 s. given that the charge of an electron is 1.6022 ✕ 10−19 c, calculate avogadro's number. assume that copper is oxidized to cu2+ ions.
Answers: 1
Do you know the correct answer?
How can u prove that there is an electric field...
Questions in other subjects:

SAT, 25.11.2021 05:30





Mathematics, 25.11.2021 05:30