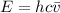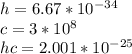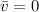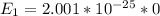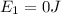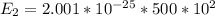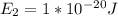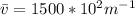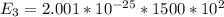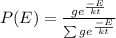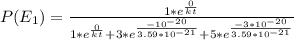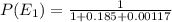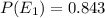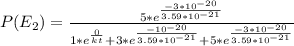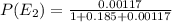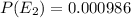Consider a molecule having three energy levels as follows:
State Energy (cm−1) Degenera...

Physics, 10.04.2020 04:54, RayOfSunsh1ne
Consider a molecule having three energy levels as follows:
State Energy (cm−1) Degeneracy
1 0 1
2 500 3
3 1500 5
Imagine a collection of N molecules all at 260. K in which one of these molecules is selected. Note: k=0.69503476cm−1⋅K−1.
1) What is the probability that this molecule will be in the lowest-energy state?2) What is the probability that it will be in the highest-energy level?

Answers: 3
Other questions on the subject: Physics


Physics, 22.06.2019 06:30, kdfawesome5582
Apebble is thrown into a calm lake, ripples are formed from the center and move outward. the water particles in the lake travel in a circular pattern that moves up and down on the surface of a lake, and the energy travels a) diagonally. b) downward. c) horizontally. d) upward.
Answers: 2

Physics, 22.06.2019 16:40, sabrinarea041904
Which are true about beta reactions? there will be more than one answer. a. it has no change b. it doesn't change the mass of the number c. it has a mass of 0.0005 atoms d. it causes transmutation.
Answers: 3

Physics, 22.06.2019 20:10, kerarucker12pe384k
Atruck with 34-in.-diameter wheels is traveling at 55 mi/h. find the angular speed of the wheels in rad/min, *hint convert miles to inches & hours to minutes:
Answers: 2
Do you know the correct answer?
Questions in other subjects:

Biology, 18.12.2019 13:31


Arts, 18.12.2019 13:31

Social Studies, 18.12.2019 13:31

Mathematics, 18.12.2019 13:31


Mathematics, 18.12.2019 13:31

Mathematics, 18.12.2019 13:31

Chemistry, 18.12.2019 13:31

Chemistry, 18.12.2019 13:31