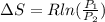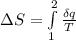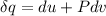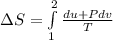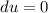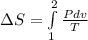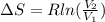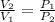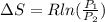A unit mass of an ideal gas at temperature T undergoes a reversible isothermal process from pressure P1 to pressure P2 while losing an amount q of heat to the surroundings at temperature T. If the gas constant of the gas is R, the entropy change of the gas âs during this process is:a. âS =R ln (P2/P1)b. âS = R ln (P1/P2)c. âS =R ln (P2/P1) - q/Td. âS = R ln (P1/P2) - q/Te. 0

Answers: 1
Other questions on the subject: Physics

Physics, 22.06.2019 02:30, JanaMiqdad1003
Agas initially at p1 = 1 bar and occupying a volume of 0.5 liter is compressed within a piston–cylinder assembly to a final pressure p2 = 4 bar. (a) if the relationship between pressure and volume during the compression is pv = constant, determine the volume, in liters, at a pressure of 3 bar. (b) repeat for a linear pressure–volume relationship between the same end states. reference
Answers: 1

Physics, 22.06.2019 03:00, coryintheswamp
What are the applications of electromagnetic waves
Answers: 1

Physics, 22.06.2019 11:00, coolfab9338
1.)the isotope cobalt-60 has a nuclear mass of 59.933820 u calculate the mass defect of cobalt-60 using the following information. mass of proton: 1.007825 u mass of neutron: 1.008665 u 1 u = 931.5 mev 2.)the isotope cobalt-60 has a nuclear mass of 59.933820 u calculate the binding energy of cobalt-60 using the following information. mass of proton: 1.007825 u mass of neutron: 1.008665 u 1 u = 931.5 mev 3.)the isotope cobalt-60 has a nuclear mass of 59.933820 u calculate the binding energy per nucleon of cobalt-60 using the following information. mass of proton: 1.007825 u mass of neutron: 1.008665 u 1 u = 931.5 mev
Answers: 3
Do you know the correct answer?
A unit mass of an ideal gas at temperature T undergoes a reversible isothermal process from pressure...
Questions in other subjects:

Mathematics, 29.01.2020 10:47




English, 29.01.2020 10:47

Mathematics, 29.01.2020 10:47

Mathematics, 29.01.2020 10:47

Mathematics, 29.01.2020 10:47

Chemistry, 29.01.2020 10:47

Mathematics, 29.01.2020 10:47