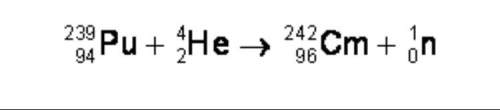
Physics, 06.03.2020 23:33, puchie1225
88 An insulated rigid tank is divided into two equal parts by a partition. Initially, one part contains 12 kmol of an ideal gas at 330 kPa and 508C, and the other side is evacuated. The partition is now removed, and the gas fills the entire tank. Determine the total entropy change during this process. 69.2 kJ/K

Answers: 2
Other questions on the subject: Physics


Physics, 22.06.2019 04:30, icantspeakengles
In a voltaic cell, electrons flow from the (positive/negative) to the (positive/negative) terminal.
Answers: 1

Physics, 22.06.2019 17:10, kaiyakunkle
What causes the development of most clouds and precipitation in the atmosphere?
Answers: 1

Physics, 22.06.2019 18:00, maren17
Air enters a gas turbine with two stages of compression and two stages of expansion at 100 kpa and 17°c. this system uses a regenerator as well as reheating and intercooling – the intercooler returns the air to the inlet temperature. the pressure ratio across each compressor is 4 ; 300 kj/kg of heat are added to the air in each combustion chamber; and the regenerator operates perfectly while increasing the temperature of the cold air by 20°c. determine the system’s thermal efficiency. assume isentropic operations for all compressor and the turbine stages and use constant specific heats at room temperature. (0.378)
Answers: 3
Do you know the correct answer?
88 An insulated rigid tank is divided into two equal parts by a partition. Initially, one part conta...
Questions in other subjects:

History, 31.07.2019 17:30


Biology, 31.07.2019 17:30



Mathematics, 31.07.2019 17:30


Mathematics, 31.07.2019 17:30

Mathematics, 31.07.2019 17:30

History, 31.07.2019 17:30