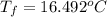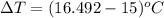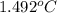Answers: 2
Other questions on the subject: Physics

Physics, 22.06.2019 14:50, MoogleCaliS
Nitrogen (n2) undergoes an internally reversible process from 6 bar, 247°c during which pν1.2 = constant. the initial volume is 0.1 m3 and the work for the process is 121.14 kj. assuming ideal gas behavior, and neglecting kinetic and potential energy effects, determine heat transfer, in kj, and the entropy change, in kj/s. show the process on a t-s diagram.
Answers: 2

Physics, 22.06.2019 16:40, Mikey5818
On your wedding day your lover gives you a gold ring of mass 3.67 g. 48 years later its mass is 3.44 g. on the average how many atoms were abrated from the ring during each second of your marriage? the atomic mass of gold is 197 u. don't enter units
Answers: 1


Physics, 22.06.2019 22:00, morganzahn16
It takes a lot of energy to get the temperature of water to increase and eventually boil because water has a high heat.
Answers: 1
Do you know the correct answer?
In a Joule experiment, a mass of 6.51 kg falls through a height of 66.8 m and rotates a paddle wheel...
Questions in other subjects:


English, 25.01.2021 20:30

Mathematics, 25.01.2021 20:30




Arts, 25.01.2021 20:30




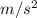 , h = 66.8 m
, h = 66.8 m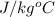
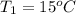
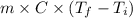
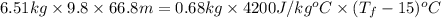
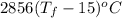
 - 15
- 15