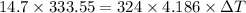A
14.7
g
ice cube is placed into
324
g
of water. C...

Physics, 14.02.2020 05:08, alonzob2367
A
14.7
g
ice cube is placed into
324
g
of water. Calculate the temperature change in the water upon complete melting of the ice. Hint: Determine how much heat is absorbed by the melting ice and then use
q
=
m
C
Δ
T
to calculate the temperature change. Use the heat of fusion for water to calculate "q"

Answers: 2
Other questions on the subject: Physics


Physics, 22.06.2019 07:00, divadebbgirl1
Examine the equation. 23490th→23088ra+42he what kind of barrier would you need to block the radioactive particles from this reaction? a. a piece of paper b. a sheet of aluminum foil c. a two-inch block of lead d. a solid concrete block
Answers: 1

Physics, 22.06.2019 10:00, kenna0
This is important 1. what happens to the speed of molecules in water vapor when it condenses to form a liquid? (2 points) the molecules slow down. the molecules speed up. the molecules slow down and then speed up. the molecules speed up and then slow down. 2. how might human activities change to compensate for a decrease in water supplies? (2 points) humans might build a factory. humans might build drainage systems. humans might extract more water from the ground. human behavior does not change because of the water cycle. i'll be grateful if anyone can answer these 2 questions !
Answers: 1

Physics, 22.06.2019 10:30, tyw3366812778
Awoman holds a book by placing it between her hands such that she presses at right angles to the front and back covers. the book has a mass of m = 1.8 kg and the coefficient of static friction between her hand and the book is μs = 0.67. no attempt 50% part (a) what is the weight of the book, fgb in newtons?
Answers: 3
Do you know the correct answer?
Questions in other subjects:

Mathematics, 18.10.2020 14:01



English, 18.10.2020 14:01


Social Studies, 18.10.2020 14:01

Mathematics, 18.10.2020 14:01


Mathematics, 18.10.2020 14:01


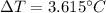 is the drop in the water temperature.
is the drop in the water temperature.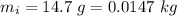 mass of water,
mass of water, 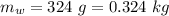
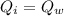
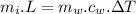 ......................(1)
......................(1) latent heat of fusion of ice
latent heat of fusion of ice 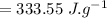
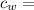 specific heat of water
specific heat of water 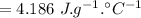
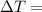 change in temperature
change in temperature