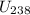Uranium-238 (U238) has three more neutrons than uranium-235 (U235). Compared to the speed of sound in a bar of U235, is the speed of sound in a bar of U238 higher, lower, or the same? There are several factors that play a role.
(a) Chemically, the atoms of these two isotopes behave in essentially identical ways, since the number of protons (92) is identical and the clouds of 92 electrons are nearly identical. The interatomic distance, and the effective "stiffness" of the interatomic bond, both depend on the chemical properties of the atoms. Therefore, which of the following statements are true?
A) The effective stiffness of the interatomic "spring" is the same in the two isotopes, since this "spring" is a model for the interactions of the outer electrons, which are the same for different isotopes of the same atom.
B) An atom with more mass in the nucleus is bigger, so the distance between neighboring atoms is larger.
C) The interatomic distance is the same for the two isotopes, because it depends on the size of the electron cloud, not the nuclear mass.
D) The effective stiffness of the interatomic "spring" is greater for U238 because the mass is greater.

Answers: 2
Other questions on the subject: Physics



Physics, 23.06.2019 00:10, alisonnn101
During cooling, the kinetic energy of the molecules falls. why does this happen? a. the motion of the molecules increases. b. the motion of the molecules slows down. c. the van der waals forces between molecules decrease d. the forces of attraction between the molecules are overcome.
Answers: 2

Physics, 23.06.2019 02:00, nativebabydoll35
Frequency of electromagnetic waves that a radio station is assigned. what do you call this type of wave?
Answers: 2
Do you know the correct answer?
Uranium-238 (U238) has three more neutrons than uranium-235 (U235). Compared to the speed of sound i...
Questions in other subjects:


Mathematics, 12.05.2021 19:00

Physics, 12.05.2021 19:00


History, 12.05.2021 19:00


History, 12.05.2021 19:00

Social Studies, 12.05.2021 19:00


Mathematics, 12.05.2021 19:00


 and
and