
How much heat energy is required to convert 93.4 g of solid ethanol at − 114.5 ° c to gasesous ethanol at 149.8 ° c ? the molar heat of fusion of ethanol is 4.60 kj/mol , and its molar heat of vaporization is 38.56 kj/mol . ethanol has a normal melting point of − 114.5 ° c and a normal boiling point of 78.4 ° c . the specific heat capacity of liquid ethanol is 2.45 j / g ⋅ ° c , and that of gaseous ethanol is 1.43 j / g ⋅ ° c .

Answers: 3
Other questions on the subject: Physics


Physics, 22.06.2019 02:00, apalacios3503
Which safety measures should you follow during a thunderstorm? check all that apply. (a) avoid touching anything that conducts electricity. (b) avoid touching a person who has been struck by lightning. (c) go outside. (d) keep your computer turned off. (e) stay out of water. (f) stay inside.
Answers: 2

Physics, 22.06.2019 05:00, masie03
Red light strikes a metal surface and electrons are ejected. if violet light is now used with a 10% greater intensity, what will happen to the ejection rate (number of ejected electrons per second) and the maximum energy of the electrons? a) greater ejection rate; same maximum energyb) same ejection rate; greater maximum energyc) greater ejection rate; greater maximum energyd) same ejection rate; same maximum energye) none of the above answers are correct
Answers: 1

Physics, 22.06.2019 07:20, hardwick744
If 2 moles of co_2 at 2l and 500k are expanded reversibly to 20l, more work can be obtained from an adiabatic process than from an isothermal process. is the above statement true or false?
Answers: 3
Do you know the correct answer?
How much heat energy is required to convert 93.4 g of solid ethanol at − 114.5 ° c to gasesous ethan...
Questions in other subjects:



Mathematics, 01.06.2020 23:59

Computers and Technology, 01.06.2020 23:59


Biology, 01.06.2020 23:59

English, 01.06.2020 23:59

Mathematics, 01.06.2020 23:59


Social Studies, 01.06.2020 23:59


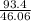 = 2.02 moles
= 2.02 moles



