
Physics, 28.12.2019 03:31, coolfreekid3
Due to tides mean sea level off of newport beach reaches a height of 1.3 meters during high tide and 0.3 meters during low tide. successive high tides occur every 12 hours (43,200 seconds). a buoy with mass m = 40 kg is floating in the ocean off of newport beach.1) relevant concepts/equations. (5 points.)2) assume we begin to measure the buoy’s displacement at high tide which occurs exactly at 12: 00 am (0 seconds). also assume we can model the buoy’s displacement as a simple undamped oscillation. what is the amplitude and phase angle for the buoy’s displacement? (10 points)3) during one half cycle of six hours (21600 seconds), the buoy’s displacement passes through an angle of 180 degrees. from this information, what is the angular frequency ω of the buoy? (5 points)4) using your previous answer, what is the force constant ‘k’ acting on the buoy? (5 points)5) what is the maximum velocity of the buoy? what is the maximum acceleration of the buoy? (10 points)6) what is the energy of the buoy due to tidal displacement? (5 points)7) how much work is done during one low tide to high tide cycle? how much power per hour is required to accomplish this? (assume g= 9.81m/s^2 , compare your answer to a 65w light bulb which uses 65 watts per hour.)

Answers: 1
Other questions on the subject: Physics

Physics, 21.06.2019 18:40, JellalFernandes
An observer that stands still as the electron moves by him, will observe
Answers: 3



Physics, 22.06.2019 07:10, PastyMexican24
1. how much energy is needed to raise the temperature of 40.0 g of argon from 25c to 40c? the specific heat capacity of argon is 0.520 j/(g·k) 2a. 23.0 ml of 0.100 m hcl (standard) are added from a buret to neutralize 50.0 ml of an unknown basic solution. 2b. if the oh- produced in the previous reaction came from ca(oh)2, then what is the molarity of the ca(oh)2? 3.calculate the new freezing-point of a solution when 60.5 grams of cacl2 solute is dissolved in 0.612 kg of water. 4. what is the maximum number of moles of alcl3 that can be produced from 5.0 mol al and 6.0 mol cl2? 5. a sample of oxygen gas has a volume of 150 ml when its pressure is 0.923 atm. if the pressure is increased to 0.987 atm and the temperature remains constant, what will the new volume be? 6. nitrogen gas in a closed container at a temperature of 100.0 oc and 3.0 atm is heated to 300 oc. what is the pressure of the gas at the higher temperature?
Answers: 3
Do you know the correct answer?
Due to tides mean sea level off of newport beach reaches a height of 1.3 meters during high tide and...
Questions in other subjects:

Biology, 11.10.2019 01:40


Mathematics, 11.10.2019 01:40

History, 11.10.2019 01:40

Mathematics, 11.10.2019 01:40

English, 11.10.2019 01:40





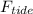 = m·a
= m·a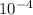 s⁻¹
s⁻¹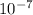 N/m
N/m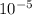 m/s
m/s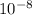 m/s²
m/s² 



