Physics, 26.12.2019 23:31, algahimnada
Light shines through atomic hydrogen gas. it is seen that the gas absorbs light readily at a wavelength of 91.65 nm. what is the level to which the hydrogen is being excited by the absorption of light of this wavelength? assume that most of the atoms in the gas are in the lowest level.

Answers: 1
Other questions on the subject: Physics

Physics, 21.06.2019 21:30, queenkimm26
Look at the potential energy diagram for a chemical reaction. which statement correctly describes the energy changes that occur in the forward reaction?
Answers: 1

Physics, 22.06.2019 12:00, ameliaxbowen7
Suppose a comet has an orbital period of 309.1 years around the sun. what is it’s average distance from the sun?
Answers: 1

Physics, 22.06.2019 20:30, cupcake3103670
Aball is thrown from the top of a building with an initial velocity of 21.9 m/s straight upward, at an initial height of 51.6 m above the ground. the ball just misses the edge of the roof on its way down, as shown in the figure. (a) determine the time needed for the ball to reach its maximum height. (b) determine the maximum height. (c) determine the time needed for the ball to return to the height from which it was thrown, and the velocity of the ball at that instant. (d) determine the time needed for the ball to reach the ground. (e) determine the velocity and position of the ball at t = 5.35 s.
Answers: 1
Do you know the correct answer?
Light shines through atomic hydrogen gas. it is seen that the gas absorbs light readily at a wavelen...
Questions in other subjects:

English, 16.11.2020 23:40


Mathematics, 16.11.2020 23:40





Mathematics, 16.11.2020 23:40


Mathematics, 16.11.2020 23:40


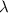 = Wavelength of the light absorbed = 91.65 nm = 91.65 x 10⁻⁹ m
= Wavelength of the light absorbed = 91.65 nm = 91.65 x 10⁻⁹ m 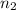 = nth level = n
= nth level = n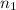 = 1
= 1