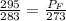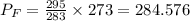Suppose you start your day in denver, on a cool 10∘c spring day. the local atmospheric pressure is 85 kpa. you fill your car's tires until the gauge shows 210 kpa (about 30 psi). you then drive up to fairplay, colorado, where the atmospheric pressure is lower-- 70 kpa --and the temperature drops to 0∘c. when you finish lunch, you check your tire pressure.

Answers: 2
Other questions on the subject: Physics



Physics, 22.06.2019 17:00, cia196785920
(a) if the pressure in gas is doubled while its volume is held constant, by what factor do (i) vrms and (ii) change? (b) is it possible to boil water at room temperature (20oc) without heating it? explain.
Answers: 3
Do you know the correct answer?
Suppose you start your day in denver, on a cool 10∘c spring day. the local atmospheric pressure is 8...
Questions in other subjects:



History, 28.02.2020 03:43


Mathematics, 28.02.2020 03:43


History, 28.02.2020 03:44

Mathematics, 28.02.2020 03:44